2022年新的慢性淋巴细胞白血病治疗方法
最后更新2023 年 11 月 13 日

你可以合法地获得新药,即使它们在你的国家没有得到批准。
了解更多 "什么是慢性淋巴细胞白血病(CLL)?
慢性淋巴细胞白血病(CLL)是一种生长缓慢的疾病,未成熟的淋巴细胞(白细胞,是人体免疫系统的细胞)过多,主要存在于血液和骨髓中。
有时,在疾病的后期,在淋巴结中发现癌细胞,这种疾病被称为小淋巴细胞淋巴瘤。白血病是一种发生在骨髓等造血组织中的癌症。它是一种不会产生肿块或肿瘤的疾病,而是以大规模产生异常和不成熟的血细胞为特征。如果是急性白血病,不成熟的血细胞会迅速增加,而慢性白血病的血细胞则会以比较正常的方式发育,导致疾病发生的时间较长。1
慢性淋巴细胞白血病(CLL)有哪些新的治疗方法?
有几种治疗慢性淋巴细胞白血病(CLL)的药物已经被批准。以下是一些最新的慢性淋巴细胞白血病(CLL)治疗方法。
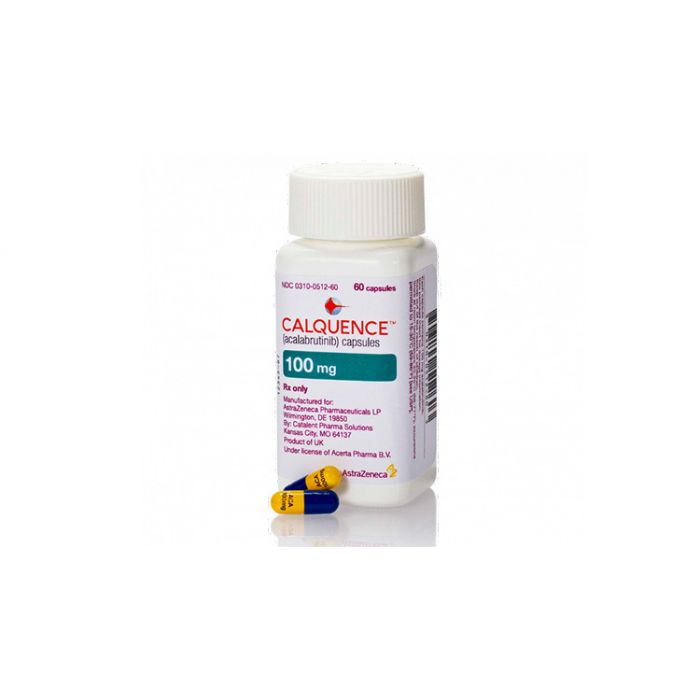
Calquence (acalabrutinib)4,5
Calquence (acalabrutinib)是一种激酶抑制剂,适用于治疗患有慢性淋巴细胞白血病(CLL)的成年患者。Calquence (acalabrutinib)可单独使用(单药治疗)于以前接受过治疗的CLL患者,也可单独使用或与obinutuzumab联合使用于以前未接受过治疗的患者。
2019年11月21日,美国食品和药物管理局(FDA)批准acalabrutinib (Calquence;AstraZeneca)作为慢性淋巴细胞白血病(CLL)或小淋巴细胞淋巴瘤(SLL)成年患者的初始或后续疗法。
2020年11月9日,欧洲医疗机构(EMA),欧盟对慢性淋巴细胞白血病(CLL)成年患者的治疗。
Imbruvica (ibrutinib)6
英博维卡(ibrutinib)是一类Bruton's酪氨酸激酶(BTK)抑制剂,用于治疗成人患有。幔细胞淋巴瘤(MCL),之前至少接受过一次治疗。慢性淋巴细胞白血病(CLL)/小淋巴细胞淋巴瘤(SLL)。
2020年4月21日,美国食品和药物管理局(FDA)批准了Imbruvica(ibrutinib)与rituximab ,用于治疗以前未经治疗的慢性淋巴细胞性白血病(CLL)或小淋巴细胞性淋巴瘤(SLL)患者。这次批准标志着Imbruvica (ibrutinib)自2013年首次获批以来的第11次FDA批准,也是第六次批准用于CLL,即成人最常见的白血病形式。
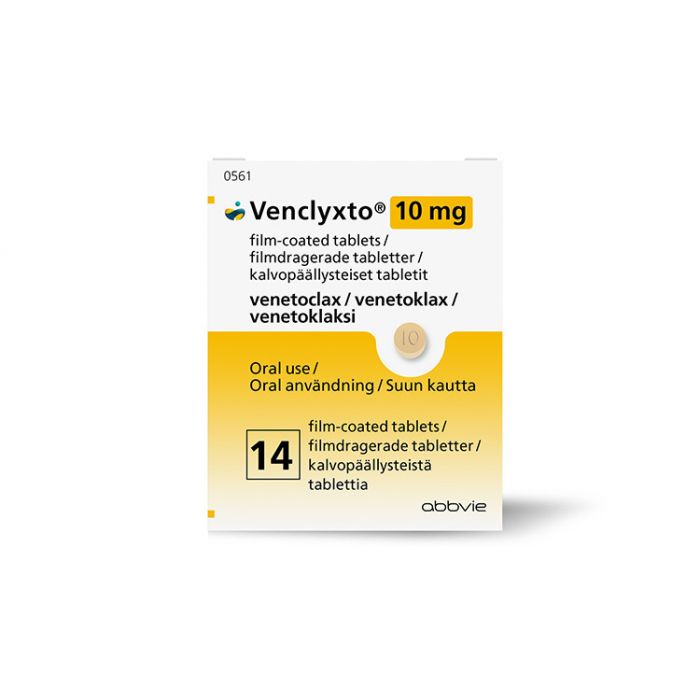
Venclyxto/Venclexta (venetoclax)7,
Venclexta/Venclyxto (venetoclax)是一种B细胞淋巴瘤-2(BCL-2)抑制剂(化疗),适用于治疗患有慢性淋巴细胞白血病(CLL)的患者,无论是否有17p缺失,之前至少接受过一次治疗。
Venclexta/Venclyxto (venetoclax) 被批准。
- 美国食品药品监督管理局(FDA):2016年4月11日,用于治疗具有或不具有17p缺失的CLL或小淋巴细胞淋巴瘤(SLL)患者,且之前至少接受过一次治疗。
- 2016年12月5日,欧盟医疗机构(EMA),用于治疗不适合使用B细胞受体通路抑制剂或已失败的成年患者存在17p缺失或TP53突变的CLL,以及用于治疗不存在17p缺失或TP53突变的成年患者化疗免疫治疗和B细胞受体通路抑制剂均失败的CLL。
- 澳大利亚治疗商品管理局(TGA),2017年1月5日,与rituximab ,用于治疗既往接受过至少一种治疗的成年CLL患者。作为单药治疗,它适用于治疗有17p缺失的复发或难治性CLL患者或没有其他合适治疗方案的复发或难治性CLL患者。
- 加拿大卫生部,2016年9月,与rituximab ,用于治疗既往接受过至少一种治疗的CLL患者。作为单药治疗,它适用于治疗既往接受过至少一种治疗的17p缺失的CLL患者,或既往接受过至少一种治疗且没有其他治疗选择的无17p缺失的CLL患者。
- 2017年11月2日,新西兰MedSafe公司与rituximab ,用于治疗既往接受过至少一种治疗的CLL患者。作为单药治疗,它适用于治疗具有17p缺失的复发或难治性CLL患者,或没有其他合适治疗方案的复发或难治性CLL患者。
如果您正试图获得在您居住的国家以外批准的慢性淋巴细胞白血病(CLL)的治疗方法,我们可能会在您的主治医生的帮助下帮助您获得该药物。您可以在下面阅读更多关于我们可以帮助您获得的药物及其价格。
为什么要通过everyone.org获得慢性淋巴细胞白血病(CLL)治疗?
everyone.org 是在荷兰海牙卫生部注册的药品批发商(注册号为 16258 G)。 我们已帮助超过 85 个国家的患者获得了数千种药品,其中包括:吲哚美辛。只要有主治医生的处方,我们的专家团队就可以安全、合法地指导您获得治疗 CLL 的新药。如果您或您认识的人希望获得在其居住地尚未获得批准的药物,我们可以为您提供支持。/contact-us 联系我们,了解更多信息。 联系我们了解更多信息。
参考文献:
- Nhs.uk
- Astrazeneca.com
- Astrazeneca.com
- Globenewswire.com
- Calquence (acalabrutinib)- Thesocialmedwork.com
- Imbruvica (ibrutinib)- Thesocialmedwork.com
- Venclyxtob (vynclyxta/venetoclax) - Thesocialmedwork.com
免责声明:本文无意影响或冲击你的主治医生所提供的护理。在没有咨询你的医疗服务提供者之前,请不要对你的治疗做出改变。本文无意于诊断或治疗疾病,也无意于影响治疗方案。everyone.org尽可能勤奋地编辑和更新本页的信息。但是,everyone.org不保证本网页所提供信息的正确性和完整性。